Chronic kidney disease (CKD) has traditionally been viewed as a disorder of blood pressure, blood sugar and kidney filtration. However, emerging research into the gut–kidney axis suggests that CKD may also be deeply connected to the health of the gut microbiome.
Could chronic kidney disease partly be a microbiome-driven condition?
According to the Centres for Disease Control and Prevention (CDC), approximately 37 million people in the United States are living with kidney disease, representing about 15% of the adult population. Alarmingly, nearly 90% of individuals with CKD are unaware they even have it. Among those with severely reduced kidney function who are not yet on dialysis, around one in two do not realise they are affected. Diabetes and hypertension remain the leading causes of CKD.
When someone is diagnosed with chronic kidney disease, attention is typically focused on kidney function markers, medications and dietary changes. Gut health and the microbiome are rarely top priorities. Yet growing evidence suggests that gut dysbiosis in CKD plays a critical role in inflammation, uremic toxin production and disease progression.
In this article, we explore the relationship between the microbiome and CKD and why supporting gut health may be an essential part of an integrative kidney support strategy.
What Is the Gut–Kidney Axis?
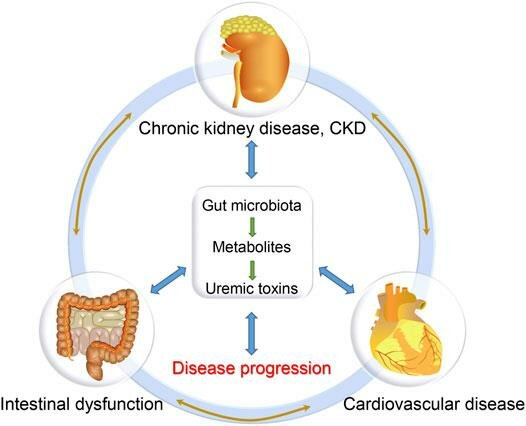
The gut–kidney axis refers to the bidirectional relationship between kidney function and the gut microbiome.
- Reduced kidney function increases circulating uremic toxins.
- These toxins disrupt gut barrier integrity.
- Gut permeability increases systemic inflammation.
- Inflammation further accelerates kidney decline.
This creates a self-reinforcing cycle in chronic kidney disease.
Supporting the microbiome may help interrupt this cycle.
Image via The-gut-kidney-heart-axis-hypothesis.jpg (526×426)
FUT2 Gene and the Microbiome in CKD
The FUT2 gene plays a key role in shaping the gut microbiota by encoding an enzyme that synthesises fucosylated sugars in the intestinal lining. These sugars act as an important food source for beneficial bacteria, particularly Bifidobacterium species.
Through a process known as fucosylation, fucosylated glycans function as prebiotics, supplying nutrients that support the growth of beneficial microbes. When this process is disrupted, the balance of the gut microbiome shifts.
Secretors vs Non-Secretors
Individuals with a functional FUT2 gene are known as secretors, meaning they release fucosylated antigens into the gut mucosa.
Those with nonfunctional FUT2 variants are called non-secretors and have reduced fucose availability on intestinal surfaces.
Non-secretors typically exhibit:
- Lower abundance and diversity of Bifidobacterium species, including B. bifidum, B. adolescentis, and B. catenulatum/pseudocatenulatum
- Reduced overall microbial diversity
- Higher levels of potentially pathogenic bacteria
- Decreased production of short-chain fatty acids (SCFAs)
Short-chain fatty acids are essential for:
- Gut barrier integrity
- Immune regulation
- Anti-inflammatory signalling
Reduced Bifidobacteria in non-secretors may increase susceptibility to dysbiosis, something particularly relevant in chronic kidney disease.
FUT2 Gene and Vitamin B12
The FUT2 gene is also crucial for the synthesis of fucosyltransferase. This enzyme plays a role in vitamin B12 absorption.
Variants in the FUT2 gene can influence:
- Microbial composition
- B12 bioavailability
- Gut-mediated nutrient absorption
Even when dietary intake is adequate, altered microbial balance may impair B12 absorption. Over time, this can contribute to deficiency, particularly in elderly individuals, vegans and vegetarians.
Symptoms of Vitamin B12 Deficiency
- Megaloblastic anaemia
- Impaired immune defence
- Elevated homocysteine
- Gastrointestinal disturbances
- Neurological dysfunction
Given the inflammatory burden already present in CKD, maintaining optimal B12 levels becomes even more important.
Do You Have Fut2 Gene Symptoms? Fut2 Gene | MthfrGeneHealth
Frontiers | Fucosyltransferase 2: A Genetic Risk Factor for Intestinal Diseases
Why Bifidobacteria Matter in Chronic Kidney Disease
Bifidobacteria play a central role in gut health and systemic regulation. In patients with CKD, their importance is amplified.
1. Reduction of Uremic Toxins
Bifidobacteria help reduce the production and absorption of uremic toxins, such as indoxyl sulphate and p-cresyl sulphate, which are known to accelerate CKD progression.
2. Reduced Inflammation
Chronic inflammation drives kidney decline. Bifidobacteria support anti-inflammatory signalling and may help lower systemic inflammatory burden.
3. Strengthening the Gut Barrier
These bacteria support tight junction integrity, reducing intestinal permeability and preventing endotoxin translocation into the bloodstream.
4. Detoxification Support
Bifidobacteria assist in nutrient metabolism and microbial detoxification pathways.
In chronic kidney disease, where gut barrier dysfunction and toxin accumulation coexist, maintaining adequate levels of Bifidobacteria is critical.
Modern Drivers of Microbiome Disruption in CKD
The microbiome is highly dynamic. It responds to environmental exposure, lifestyle, diet, infections, medications and physiological stressors.
Over recent decades, significant shifts in microbiome composition have occurred globally.
Electromagnetic Fields (EMFs)
Exposure to phones, Wi-Fi, and power lines has been studied in experimental models. Some animal studies suggest that microbiome shifts may occur after long-term exposure, potentially mediated by oxidative stress pathways.
While human data is still emerging, oxidative stress is a known contributor to both dysbiosis and CKD progression.
The response of human bacteria to static magnetic field and radiofrequency electromagnetic field | Journal of Microbiology | Springer Nature Link
EMF Sensitivity and Digestion: Examining the Evidence and Protective Strategies – GutNow
Effects of electromagnetic fields exposure on the antioxidant defense system – PMC
COVID-19 Infection
SARS-CoV-2 infection has been shown to:
- Reduce microbiome diversity
- Decrease beneficial bacteria, including Bifidobacterium
- Increase inflammatory-associated species
These alterations may persist for months in some individuals.
Alterations in Gut Microbiota of Patients With COVID-19 During Time of Hospitalization – PubMed
Psychological Stress and the Gut–Brain Axis
Chronic stress activates the hypothalamic–pituitary–adrenal (HPA) axis, increasing cortisol and sympathetic activity.
This affects:
- Gut motility
- Intestinal permeability
- Immune signalling
Stress-induced dysbiosis may indirectly worsen chronic kidney disease through inflammatory pathways.
Antibiotics and Medications
Antibiotics reduce microbial diversity and can permanently alter gut ecology after repeated use.
Other medications known to influence microbiota composition include:
- Proton pump inhibitors (PPIs)
- NSAIDs
- Metformin
- SSRIs
Medication-induced dysbiosis may contribute to CKD progression via uremic toxin production.
Modern Diet and Fibre Depletion
The modern diet is typically:
- Low in fibre
- High in processed foods
- High in refined sugars
- High in industrial seed oils
Low fibre intake reduces short-chain fatty acid production, weakening gut barrier integrity and decreasing Bifidobacteria abundance.
Mould, Mycotoxins and Oxalates: Connecting the Dots in CKD
Environmental exposures interact with genetic susceptibility and microbial balance particularly in individuals with chronic kidney disease.
Mould and Mycotoxins
Mycotoxins may impair kidney function through:
- Oxidative stress
- Inflammation
- Direct tubular toxicity
Chronic inflammation is a driver of fibrosis in kidney tissue. In high-exposure scenarios, environmental toxins are considered potential contributors to CKD progression.
For example, deoxynivalenol (DON), produced by Fusarium species, has demonstrated renal tubular toxicity in experimental models.
Candida, Mould and Oxalates
Candida and mould species can produce oxalates. Dysbiosis may impair oxalate breakdown.
This means that even a low-oxalate diet may not prevent elevated oxalate levels if microbial imbalance persists.
A high oxalate burden, combined with gut permeability, may further increase inflammatory stress in CKD.
Restoring Bifidobacteria in Chronic Kidney Disease
Protecting and restoring Bifidobacteria is an important strategy in reducing inflammation and toxin load in CKD.
Image via Synbiotic Diets: The Combined Power of Probiotics and Prebiotics for Gut Health
Strategies include:
Dietary and Lifestyle Foundations
- Limit processed foods
- Avoid unnecessary antibiotics
- Address constipation
- Increase dietary fibre
- Include fruits and vegetables (as clinically appropriate for CKD)
- Engage in regular physical activity
Probiotics
Supplementation with Bifidobacterium-containing probiotics may help restore microbial balance.
Prebiotics
Prebiotic fibres such as:
- Inulin
- Fructooligosaccharides (FOS)
- Galactooligosaccharides (GOS)
- Resistant starch
Synbiotics
Combining probiotics and prebiotics enhances colonisation.
Synbiotics enriched with polyphenols have shown promise in improving:
- Gut health
- Metabolic function
- Immune regulation
Bifidobacterium in Kidney Disease and Other Human Diseases
Is Chronic Kidney Disease a Microbiome Disease?
Modern living has reshaped the human microbiome through diet, stress, infections, environmental exposures and medications.
Chronic dysbiosis has been associated with:
- Metabolic syndrome
- Autoimmune disorders
- Cardiovascular disease
- Chronic kidney disease
Research increasingly links gut microbial imbalance to CKD progression via:
- Uremic toxin production
- Systemic inflammation
- Immune dysregulation
- Barrier dysfunction
Genetics (such as FUT2 status), oxalate burden and environmental mould exposure may compound microbiome instability in CKD.
The question may no longer be whether the microbiome influences chronic kidney disease, but how much of CKD progression is driven by gut dysfunction.
As research into the gut–kidney axis expands, supporting microbial balance may become one of the most important integrative strategies in slowing kidney decline.
Modern-day living has slowly reshaped the human gut microbiome through multiple factors. Highly processed, low-fibre diets reduce beneficial bacteria such as Bifidobacterium and decrease short-chain fatty acid production, weakening gut barrier integrity and promoting systemic inflammation. Chronic psychological stress alters gut motility, immune signalling, and intestinal permeability via the gut–brain axis, further contributing to dysbiosis. Widespread antibiotic use and repeated infections, including COVID-19, have also been shown to disrupt microbial diversity.
Chronic dysbiosis has been associated with inflammatory conditions such as metabolic syndrome, autoimmune disorders, CKD and cardiovascular disease. Research links gut microbial imbalance to chronic kidney disease through mechanisms involving uremic toxin production, systemic inflammation and altered immune regulation.
For individuals with CKD, supporting healthy levels of Bifidobacteria is especially important because kidney impairment and gut imbalance reinforce each other in a harmful cycle. Reduced kidney function increases uremic toxins, which disrupt the intestinal barrier and shift the microbiome toward toxin-producing bacteria. Healthy amounts of Bifidobacteria help counter this by strengthening gut barrier integrity, lowering inflammation, reducing uremic toxin production and supporting immune balance.
Genetics also play a role. Variants of the FUT2 gene influence the secretion of fucosylated glycans in the gut, which serve as a food source and attachment sites for Bifidobacteria. Individuals with non-secretor FUT2 variants often show reduced Bifidobacteria abundance and may be more vulnerable to dysbiosis, something we would like to avoid in CKD.
High oxalate exposure and chronic exposure to mould toxins can disrupt gut microbial diversity.
Altogether, a genetic susceptibility, oxalate burden, and environmental mould exposure can all compound microbiome instability in CKD. Protecting and restoring Bifidobacteria is an important consideration in helping to reduce toxin load, inflammation and gut-related complications in kidney disease.
Frequently Asked Questions About CKD and the Microbiome
Can gut dysbiosis cause chronic kidney disease?
Emerging evidence suggests that gut dysbiosis may contribute to CKD progression by increasing uremic toxins and systemic inflammation.
What bacteria are reduced in CKD?
Bifidobacterium species are frequently reduced in patients with chronic kidney disease.
What is the gut–kidney axis?
The gut–kidney axis describes the bidirectional relationship between kidney function and the gut microbiome.
As always, before making any changes to your diet or supplementation, speak to your trusted healthcare provider.